Atoms
Celestial Bodies
- Space Travel Equipment
- Stars
- Rotation and Revolution
- Relation Between Escape Velocity And Orbital Velocity
- Dwarf Planets
- Difference Between Solar Eclipse And Lunar Eclipse
- Difference Between Equinox And Solstice
- The Escape Velocity Of Earth
- Solar System
- Difference Between Stars And Planets
- Difference Between Asteroid And Meteoroid
- Constellations
Circuits
电路 (diàn lù)
电路 (Diànlù)
电路
通信系统Pdf
二极管
地球科学
电荷
电
- 类型的齿轮
- 电子产品在日常生活中
- 类型的汽车
- 类型的直流电机
- 类型的交流电机
- 晶体管工作
- 转矩电流环
- 电动机
- 电阻温度依赖性
- Rms值交流电
- 电抗和阻抗
- 相量表示法交流
- 平行板电容器
- 焦耳定律
- 电力
- 磁场对载流导线的影响
- 电流密度
- 导体绝缘体
- 导电
- 碳电阻器
- 直流发电机
- 类型的发电机
- 类型的电流
- 直流发电机类型
- Torque On Dipole
- 电流的热效应
- 电动发电机
- 静电
- 电阻率不同的材料
- 电场的物理意义
- 介电常数和磁导率
- 电能和权力
- 电流在导体
- 电动汽车
- 位移电流
- 电阻与电阻率之间的差异
- 电动机和发电机之间的区别
- 接地和接地之间的区别
- 电流线圈
- 水的电导率
- 导电的液体
Electricity
电磁波
电磁
静电学
能量
- 能量
- 能源类型
- 热能
- 太阳能项目
- 太阳能汽车
- Ev和Joule之间的关系
- 动能和完成的功
- 能量转换
- 一维和二维的弹性和非弹性碰撞
- 常规能源和非常规能源
- 太阳能炊具
- 潮汐能
- 能源
- 太阳能和光伏电池
- 动能与动量的关系
- 热量与焦耳的关系
- 能源及其对环境的影响
- 能源考虑
流体
武力
Force
摩擦
万有引力
热
动力学理论
光
- 镜面反射漫反射
- 人眼
- 结构人眼功能
- 阴影的形成
- 反射和折射之间的区别
- 相干源
- 光的透射、吸收和反射
- 透明半透明和不透明
- 阳光白色
- 单狭缝衍射
- 拉曼散射
- 粒子自然光光子
- 真实图像与虚拟图像的区别
- 衍射和干涉的区别
磁性
运动
- 运输历史记录
- 速度-时间图
- 旋转动能
- 刚体和刚体动力学
- 扭矩和速度之间的关系
- 粒子的直线运动
- 周期性运动
- 动量和惯性之间的差异
- 动量守恒
- 运动测量类型
- 扭矩
- 慢速和快速运动
- 滚动
- 刚体平移运动和旋转运动
- 相对速度
- 径向加速度
- 速度和速度之间的区别
- 动力学和运动学的区别
- 连续性方程
- 线性动量守恒
自然资源
核物理学
光学
Optics
- Reflection of Light and Laws of Reflection
- Concave Lens
- Total Internal Reflection
- Thin Lens Formula For Concave And Convex Lenses
- Spherical Mirror Formula
- Resolving Power Of Microscopes And Telescopes
- Refractive Index
- Refraction Of Light
- Refraction Light Glass Prism
- Reflection On A Plane Mirror
- Reflection Lateral Inversion
- Rainbow
- Photometry
- Difference Between Simple And Compound Microscope
- Difference Between Light Microscope And Electron Microscope
- Concave Convex Mirror
- Toric Lens
- The Lens Makers Formula
- Simple Microscope
Oscillation
Pressure
- Thrust Pressure
- Relation Between Bar And Pascal
- Regelation
- Sphygmomanometer
- Relation Between Bar And Atm
- Difference Between Stress And Pressure
Quantum physics
- Quantum physics
- Rydberg Constant
- Electron Spin
- Casimir Effect
- Relativity
- Quantum Mechanics
- Electrons And Photons
Radioactivity
- Relation Between Beta And Gamma Function
- Radioactivity Beta Decay
- Radioactive Decay
- Stefan Boltzmann Constant
- Radioactivity Gamma Decay
- Radioactivity Alpha Decay
- Radiation Detector
Scalars and Vectors
- Scalars and Vectors
- Triangle Law Of Vector Addition
- Scalar Product
- Scalar And Vector Products
- Difference Between Scalar And Vector
Scientific Method
- Scientific Methods
- Safety Measures Technology
- Difference Between Science And Technology
- Scientific Investigation
Semiconductors
- Semiconductor Devices
- Junction Transistor
- Semiconductor Diode
- Difference Between Npn And Pnp Transistor
Solid Deformation
- Solid State Physics
- Solid Deformation
- Stress
- Shear Modulus Elastic Moduli
- Relation Between Elastic Constants
- Elastic Behavior Of Solids
- Tensile Stress
- Stress And Strain
- Shearing Stress
- Elastomers
- Elastic Behaviour Of Materials
- Bulk Modulus Of Elasticity Definition Formula
Sound
- Sound waves
- Timbre
- Speed Of Sound Propagation
- Sound Waves Need Medium Propagation
- Sound Reflection
- Sound Produced Humans
- Doppler Shift
- Difference Between Sound Noise Music
- The Human Voice How Do Humans Create Sound With Their Vocal Cord
- Sound Vibration Propagation Of Sound
- Sound Produced Vibration Object
- Reverberation
- Doppler Effect
System of Particles and Rotational Dynamics
Thermal Properties of Matter
- Thermal Properties of Materials
- Thermal Stress
- Thermal Expansion Of Solids
- Thermal Conductivity Of Metals
Thermodynamics
- Statistical Physics
- SI Units List
- Statistical Mechanics
- Reversible Irreversible Processes
- Carnots Theorem
- Temperature
- Kelvin Planck Statement
- Difference between Isothermal and Adiabatic Processes
Units and measurements
- Density of Air
- The Idea Of Time
- Difference Between Pound And Kilogram
- Difference Between Mass And Volume
- Dimensional Analysis
- Density Of Water
- Time Measurement
- Standard Measurement Units
- Relation Between Kg And Newton
- Relation Between Density And Temperature
- Difference Between Mass And Weight
Waves
- Space Wave Propagation
- Sharpness Of Resonance
- Relation Between Group Velocity And Phase Velocity
- Relation Between Amplitude And Frequency
- Periodic Function
- P Wave
- Destructive Interference
- Transverse Waves
- Travelling Wave
- Standing Wave Normal Mode
- S Waves
- Relation Between Frequency And Velocity
- Reflection Of Waves
- Phase Angle
- Period Angular Frequency
Work, Energy and Power
- Derivation Of Work Energy Theorem
- Conservation Of Mechanical Energy
- Relation Between Work And Energy
- Destruction Caused Cyclones
Physics Experiments
- Determine Resistance Plotting Graph Potential Difference versus Current
- To find the weight of a given Body using Parallelogram Law of Vectors
- To study the variation in volume with pressure for a sample of air at constant temperature by plotting graphs between p and v
- To measure the thickness of sheet using Screw Gauge
- To find the value of V for different U values of Concave Mirror find Focal Length
- To find the Surface Tension of Water by Capillary Rise Method
- To find the Resistance of given wire using Metre Bridge and hence determine the Resistivity of its Material Experiment
- Determine Mass of Two Different Objects Using Beam Balance
- Tracing the path of the rays of light through a glass Prism
- Tracing path of a ray of light passing through a glass slab
- Tornado Bottle
- To find image distance for varying object distances of a convex lens with ray diagrams
- To find force constant of helical spring by plotting a graph between load and extension
- To find focal length of concave lens using convex lens
- To find effective length of seconds pendulum using graph
- To find downward force along inclined plane on a roller due to gravitational pull of the earth and its relationship with the angle of inclination
- To draw the IV characteristic curve for p n junction in forward and reverse bias
- To determine Young’s modulus of elasticity of the material of a given wire
- To determine the internal resistance of a given primary cell using a potentiometer experiment
- To determine the coefficient of viscosity of given viscous liquid by measuring terminal velocity of given spherical body
- To determine specific heat capacity of given solid by method of mixtures
- To determine radius of curvature of a given spherical surface by a Spherometer
- Scope and Excitement of Physics
- Rocket science
- Relationship between frequency and length of wire under constant tension using Sonometer
- To determine equivalent resistance of resistors when connected in series and in parallel
- To convert the given galvanometer of known resistance and figure of merit into a voltmeter of desired range and to verify the same experiment
- To determine minimum deviation for given prism by plotting graph between angle of incidence and angle of deviation
- To compare the emf of two given primary cells using potentiometer experiment
Introduction
热力学是一项建立热量和功之间关系的研究。为了更好地理解它,必须深入挖掘熵和焓这两个术语的概念。本文将有序地讨论熵和焓的基本定义以及它们之间的区别。简单地说,熵是随机性的度量;另一方面,焓表示系统的总热量。
What is Entropy?
熵是一种可测量的物理性质,表示系统的无序程度。任何一种物质或介质——流体或固体,都是由分子组成的。分子越是无序和随机,熵就越大。因此,热量中可以转化为功的部分不会太多。这样,我们可以说熵是热量的函数。最初,这个概念是用“热势”这个名字创造的。后来,克劳修斯在他关于热力学第二定律的陈述中,将其定义为可逆过程中热量的微小变化与瞬时温度的比值。
$$mathrm{ΔS=frac{ΔQ}{T}}$$
如果分子的随机性较小,熵的变化也会较小。因此,固体通常比流体具有更小的熵。系统的熵,加上周围的熵,总是在增加。这意味着宇宙的熵总是在增加。
Examples
在我们周围可以看到熵变化的各种例子。在日常生活中,我们看到的证据表明,宇宙总是倾向于熵增加的方向,即随机性。日常生活中的一些例子可以列出如下-
渗透−当我们在一个封闭的平静房间里点燃一根香时,产生的烟雾总是会散开。随着分子获得随机性和无序性,它会自行扩展。这是熵增加的一个例子。我们永远不会看到烟雾在一个单独的地方沉淀和集中。
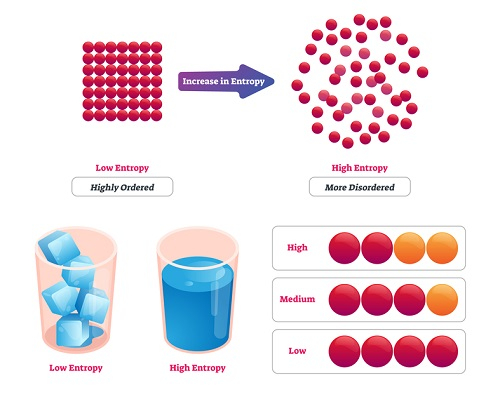
溶解也会导致熵的增加。固体实际上处于一种非常有序的状态,一旦溶解,就会进入一种更加无序的状态。当系统的随机性增加时,将糖溶解在水中会增加系统的能量,因此熵也会增加。
篝火也是熵的一个例子。燃料通常是实木、纸张或稻草,燃烧后会变成杂乱无章的灰烬。此外,还会释放烟雾和二氧化碳等各种气体。原子以膨胀的形式展开,无序度越来越大,因此据说熵增加了。
从一种状态到另一种状态的相变过程也带来了熵的变化。固态冰块在融化过程中比水具有更大的有序性,因此熵也更大。宇宙本身永远不会推动冻结的过程,即熵的减少。
What is Enthalpy?
焓是热力学系统的性质,它表示系统中的总热变化。根据热力学第一定律,它是内能与压力和体积的乘积之和。了解化学反应中焓的变化是非常重要的。由于内能中存在一些未知参数,我们无法直接测量系统的总焓。相反,我们测量焓的变化,以便更好地理解这个过程。从数学上讲,它可以表示为
$$mathrm{H=U+PV}$$
这里,U是内能,P是压力,V是系统的体积。此外,测量焓的变化也有助于我们弄清楚反应是吸热(吸收热量)还是放热(释放热量)。另一个需要注意的重要事项是,反应步骤的顺序或反应步骤的数量不会影响反应的焓变值。
Examples
焓在现实生活中有很多应用和例子。其中一些可以列出如下-
食品品牌和行业通过打破体内葡萄糖的结合来计算食物释放的能量,从而检查出售的食物中的卡路里数量。
汽车行业也会检查发动机消耗了多少能量来消耗一定量的燃料。简单地说,他们利用焓和能量变化为汽车做出高效的能源选择并节省资金。
冰箱压缩机也应用焓值。压缩机中的制冷剂化学物质被蒸发,因此,热量在吸热反应中被吸收。
Relation between Entropy and Enthalpy
在上面讨论的定义的帮助下,术语熵和焓的变化可以关联如下
当焓的变化为负时:$mathrm{Delta}$H=-ve,我们说热量被给予周围环境,即放热反应。它是一个稳定的系统,因此是自发的。这意味着周围环境的熵也会增加。
然而,当焓的变化为正时:$mathrm{Delta}$H=+ve,我们说热量是从周围添加到系统中的,即吸热反应。这意味着周围环境的熵减少了。
为了将反应的自发性与H和S完美地联系起来,使用了以下关系式
$$mathrm{ΔG=ΔH-TDelta S}$$
This equation is known as the Gibbs Helmholtz equation. In this equation, $mathrm{Delta}$G is the change in free energy. For any spontaneous reaction to happen, $mathrm{Delta}$G is always negative.
Difference between Entropy and Enthalpy
| Sr. No. | Entropy | Enthalpy |
|---|---|---|
| 1 | It is a thermodynamic measurable property | It is a kind of energy |
| 2 | It is the measure of randomness | It is the measure of the total heat content of the system |
| 3 | A system always favours the maximum value of entropy | A system always favors the minimum value of enthalpy |
| 4 | Its unit is $mathrm{JK^{1}}$ | Its unit is J$mathrm{mol^{1}}$ |
| 5 | It is denoted by symbol S | It is denoted by symbol H |
Conclusion
焓和熵是热力学中的重要术语,在检查反应的有利性和自发性时,它们密切相关。还可以看到许多差异,而焓是热含量的衡量标准,熵是系统的随机性程度。
FAQs
问题1.什么是赫斯定律
根据Hess恒定热量求和定律的说法,无论一个反应有多少步,或者这些步骤的顺序是什么,反应的总焓变都是每个步骤单独计算的所有焓变的总和。
Q2.影响反应焓变化值的两个主要因素是什么
答:反应物的温度和物质的相。物质的不同相会产生具有特定化学配方的反应物,在经历相变过程后,该反应物不一定以相同的方式传递热量。
Q3.为什么宇宙的熵在增加
答:在每一刻,大自然都在扩张,它的能量也在增加。熵是能量和随机性增加的度量,能量每时每刻都在膨胀和扩散。因此,自然是自发的,宇宙的熵也在增加。
Q4.什么是绝对熵
绝对熵是指当系统从绝对零度温度上升到某个更高的温度值时,熵的变化。
问题5.你如何知道一个反应是否有利
答:当$mathrm{Delta S}$为正时,即熵增加,这意味着系统变得更加无序,这是发生反应的有利条件。但是,当反应的$mathrm{Delta S}$为负时,熵会降低,这不是反应发生的自然有利条件。
